2019-11-07 Rongda test 2373 times
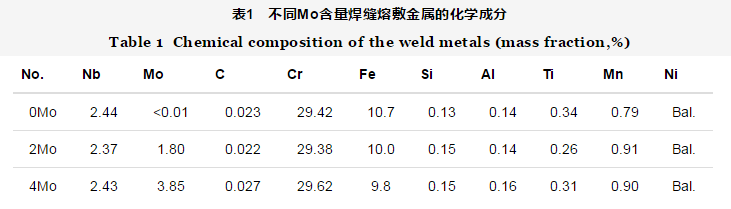
The steam generator is the key component connecting the primary and secondary circuits in PWR nuclear power plant. Its reliable service is of great significance for the safe operation of nuclear power plant. The heat transfer tube in the steam generator is welded with the tube plate, and the current welding material is nickel base corrosion-resistant alloy [1,2]. The welding joint is the weak link of the whole structure, so the quality requirements are very strict. The heat transfer tube welding of steam generator has high sensitivity of high temperature plastic loss crack and high repair rate. The superplastic crack at high temperature is a kind of intergranular crack caused by the plastic loss of austenite based materials at high temperature [3,4]. Because of its small size, difficult to detect and strong concealment, the potential damage is very great. 40% of the unplanned shutdown or suspension of nuclear power plant is caused by the failure and fracture of heat transfer tube of steam generator [5], and corrosion is the main reason for the failure of heat transfer tube [6]. The heat transfer tube material developed from the original 18-8 austenitic stainless steel to the current nickel based high Cr alloy. In this optimization process, the corrosion resistance of the material is continuously improved. Currently, the nickel base Alloy 690 (UNS n06690) has comprehensive mechanical properties and excellent corrosion resistance [7]. Because the weld metal must undergo a special welding thermal cycle, its corrosion resistance is particularly important. Ernicrfe-13 (FM 52mss), a new type of nickel base welding material, is a kind of nickel base welding material with high Cr content. Different from ernicrfe-7a (FM 52m), which is widely used in nuclear power industry, a certain amount of trace element Mo [8,9] is added to the alloy. The researchers investigated the effect of Mo element on the mechanical properties of nickel based weld material from the perspective of high temperature crack resistance, and found that this kind of weld material has a higher ability of high temperature crack resistance [10,11]. However, there are few reports on the corrosion resistance of FM 52mss. It is necessary to study the effect of Mo on the corrosion resistance of the new nickel base welding material The influence of Mo element on the corrosion resistance of corrosion-resistant alloy has been reported by scholars at home and abroad, but the conclusion is not uniform. Bogdan et al. [12] believe that increasing Mo content can reduce the cracking tendency of passivation film and improve the resistance of austenitic stainless steel to pitting corrosion, crevice corrosion and other local corrosion. Li Xiaoyan et al. [13] studied the corrosion resistance of the weld metal of Hastelloy C-276, and found that Mo element in the oxidizing medium made the TCP phase easy to be corroded, while the precipitation phase of TCP type on the grain boundary increased the chemical composition and structure inhomogeneity between the grain boundary and the grain, and increased the sensitivity of the intergranular corrosion of the weld metal. Wu Dongjiang et al. [14] studied the corrosion performance of C-276 / 316L dissimilar welded joint, and thought that the segregation of Mo element promoted the dendrite corrosion of weld structure in acid and neutral solution. It can be seen that the addition of Mo has a significant effect on the local corrosion behavior of weld metal. In this paper, the local corrosion behavior of deposited metal with different Mo content in nitric acid solution is studied, and the influence of Mo element on the local corrosion sensitivity of weld metal, such as pitting corrosion, intergranular corrosion and dendrite corrosion, is analyzed Based on the composition of fm52mss welding wire, three kinds of Ni based welding wires with different Mo content were designed and prepared, which were labeled 0mo, 2Mo and 4mo respectively. The wire diameter is about 1.2 mm The experiment of multi-layer and multi pass hardfacing was carried out by using semi-automatic cold wire filled TIG welding. The base metal is low alloy steel Q235, with the dimension of 500 mm × 125 mm × 25 mm. 22.5 ° single V groove is machined along the long side. In order to prevent the dilution of the base metal and influence the evaluation of the corrosion resistance of the weld metal during multi-layer and multi-layer surfacing, we 152 electrode was used to overlay a 7 mm thick isolation layer on the groove of Q235 steel plate Assemble two Q235 test plates with isolation layer to V-groove for filling and surfacing of deposited metal, as shown in Figure 1. Welding parameters: voltage: 10.5 ~ 11.0 V; current: 160 ~ 240 a; wire feeding speed: 900 ~ 1100 mm / min; traveling speed: 85 ~ 110 mm / min; gas protection: 99.999% pure argon; gas flow rate: 20 L / min; duty cycle: 50%. In order to reduce the tendency of hot cracking of weld metal, the interlayer temperature of weld metal is controlled below 100 ℃. The composition of three kinds of weld deposited metal is shown in Table 1. According to the RCC-M 2007 standard, the weld metal was annealed after stress removal. The heat treatment process was: 620 ℃ in muffle furnace for 29 hours and then air cooling.
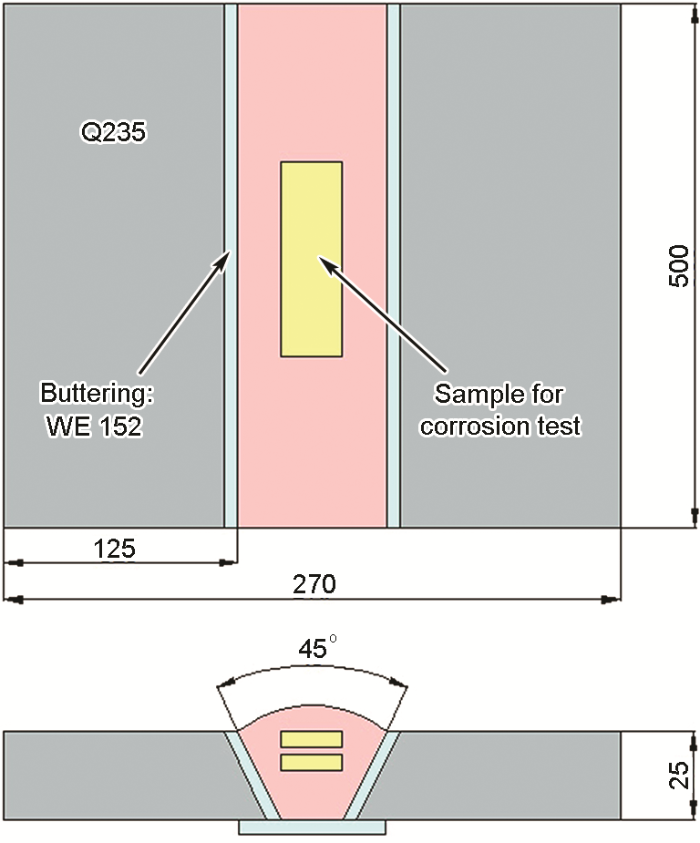
Figure 1 Schematic diagram and sampling position of welding joint
The samples were taken from the center of the deposited metal cross section by WEDM. The samples were ground by 150 × 400 × 800 × 1200 × 2000 × sandpaper and polished by 2.5 μ m diamond polishing paste. After that, the surface of the sample was electrolytic corroded in 10% chromic acid aqueous solution. The electrolytic parameters were: DC voltage 5 V, corrosion time 30 ~ 60 s. After electrolytic corrosion, 10% oxalic acid aqueous solution was used to clean the surface of the sample to remove the electrolytic corrosion products attached to the surface. The microstructure of the deposited metal with different Mo content was observed by metallographic microscope and scanning electron microscope. The composition of precipitated phase in the weld structure was analyzed by EDs. In order to determine the types of precipitates in the weld, 7% CH3OH + 93% HCl electrolyte was used to extract the precipitates in the weld, and the electrolytic current was 0.36 a. After extraction, the precipitates were dried and analyzed by XRD Conduct corrosion test according to the relevant provisions of nitric acid test in ASTM a262-2015. Take samples along the longitudinal direction of the weld with the method of electric spark cutting. Take two samples of each weld metal, with the size of 33 mm × 11 mm × 4 mm, as shown in Figure 1. After grinding and polishing the sample surface, soak it in a conical flask containing 65% nitric acid solution at 117 ℃ for 48 hours. In order to avoid interference between samples, soaking experiments were carried out separately for samples of different components and different states. In order to ensure the reliability of the experiment, the ratio of the volume of nitric acid solution to the surface area of the sample is kept above 20 ml / cm2. The mass of the sample before and after the experiment was measured with an analytical balance to the accuracy of 0.0001 G. Weight loss method was used to evaluate the corrosion rate of deposited metal samples. The average value of corrosion rate of two samples with the same composition in the same state is taken as the corrosion rate of weld deposited metal. The corrosion morphology of the sample was observed by scanning electron microscope The content of the article comes from the Internet. If there is any infringement, please contact the editor. Rong DA has a group of professional and technical personnel who have made remarkable achievements in the industry. They have a deep understanding of the testing needs in the industry and rich testing experience. In line with the principle of excellence, technical teams in different fields are established for different samples for analysis and experiment, and the senior engineer with the most professional experience is the person in charge. Ensure the accuracy and preciseness of each report. It is applicable to the relevant research and test of iron and steel enterprises, petrochemical industry, scientific research institutes, colleges and universities, etc.
Last article: NO!